Secondary Use
of health data
Secondary use of health data refers to a regulatory framework that supports using health data for other purposes than it was originally created and collected for. Currently, different countries and the European Union (EU) are working on putting in place common rules for these activities.
The harmonization of health data frameworks among different countries is a cornerstone for the digital health revolution. This sounds like an overstatement, but the purpose of the text below is to convince you that it is true.
The current main challenges are lack of rules related to the secondary use of health data and harmonization among the rules when those already exist. In the European Union, there is a common goal to enable secondary use of health data within the member states but also cross-border sharing of health data. Even in the EU the main responsibility of setting the suitable policies and implementing the right kind of data governance and data systems is with the individual countries.
For the companies operating in the health data sector, such as RemedyBytes, it is crucial that these policies and processes are put in place as quickly as possible to allow innovation and implementation of better healthcare solutions in the current landscape where there is a rapidly growing cost pressure.
What is secondary use of health data?
Let’s answer the most important question first: what is meant by secondary use of health data? Secondary use of health and social data means that the customer and register data created during health and social service sector activities will be used for purposes other than the primary reason for which they were originally saved.
The website dedicated to policy on secondary use of health data by Open Data Institute (ODI), which is a non-profit founded by the World Wide Web inventor Tim Berners-Lee, provides a great answer to this question and other useful definitions that are important for understanding the context of the topic.
Health data is a term used to describe all the information generated through the process of delivering healthcare to populations, including disease registries, public health surveys, clinical trial data, insurance claims and electronic health records.
Primary use of health data is when health data is used to deliver healthcare and to make decisions about the care of the individual from whom it was collected.
And finally, secondary use of health data is the use of the health data for anything else that is not primary use. This could be for example:
- Scientific research
- Statistics
- Development and innovation activities
- Steering and supervision of authorities
- Planning and reporting duties by authorities
- Teaching
- Knowledge management
Why do we need secondary use of health data?
In short, the idea is that the health data could be reused, anonymised and aggregated, to improve people’s health and experiences, for example by enhancing personalized healthcare, to create more efficient healthcare systems, and to foster innovation.
Secondary use of health data enables a wide range of use cases and benefits across the entire healthcare system, the benefits including:
- Optimizing health service delivery
- Reducing health inequalities through better allocation of resources
- Helping to enhance personalized healthcare
- Increase innovation
Secondary use of health data can increase the value of currently collected data from clinical settings and information collected from other data sources. The other sources of health data can be for example from registries, sickness and insurance claims records and from devices and wearable technologies. This “other data” is often referred to as real-world data (RWD).
Benefits of secondary health data
While secondary use of health data is not a silver bullet that will solve all the issues in healthcare, there are several suggestions how it could be beneficial in tackling the long list of problems in the healthcare space. Some of these benefits have been listed in the ODI’s 2021 report on Secondary use of health data in Europe which was also used to put together the benefits described in more detail below. These key benefits are:
1) Optimizing health systems
2) Improving the patient journey
3) Encouraging patient-public participation
4) Expanding innovation
While we cannot offer a specific data or reasoning why exactly these benefits have been selected, or to be more exact, why the benefits have been categorized in this manner, it should be clear that the idea is that with a better access to data and improved ways of working with the data, it is possible, at least in theory, to improve all aspects of healthcare.
Optimizing health systems
Secondary use of health data for analytics and management can reduce healthcare costs. The expectation is that better data governance allows the secondary use of health data from within the organization and other similar healthcare organizations globally will increase planning capabilities and more-efficient allocation of resources.
Delivering data-driven digital health technologies sustainably at scale would allow more equal prioritization in healthcare organizations. These tools might have the potential to provide everyone, everywhere, with equitable access to expert-level care, narrowing the global health and wellbeing gap.
In addition to being able to prioritize and equalize the healthcare system, one of the earliest motivations to improve secondary use of health data has been to modernize reimbursement and pricing models. The ability to access and analyze metadata is crucial for the value-based healthcare thinking and for the new types of pricing models, such as pay-for-performance.

Improving the patient journey
Secondary use of health data enables insights for managing people’s health, early diagnosis, prevention and healthy living. National electronic health record systems controlled by the patient are becoming increasingly common.
For example, during a pandemic, data stored in a national Electronic Health Record (EHR) system could be used by health authorities to identify persons with a particular health risk and these persons could be informed with individualized instructions.


Another important benefit of secondary use of health data is the improvement of the patient journey. Better data analysis would allow early, personalized and advanced diagnostics, personalized care pathways and support for clinical decisions, rapid access to personalized interventions and remote monitoring and care through digital health apps and tools.
Rephrasing that in a nutshell: the patient would receive an earlier and more personalized diagnosis. They would receive better care tailored to their individual needs. Their health status could be monitored in more detail and for a longer period of time.
Encouraging patient-public participation
One of the key aspects of secondary use of health data would be to allow the individual to contribute personal data, if they choose to do so. They could also opt out, if they would not want to provide their data for secondary use.
The authorities could use RWD to discuss health for educational purposes but also to inform decision-makers with better data.

Better use of RWD could reduce some of the challenges that were seen during the COVID-19 pandemic, where it sometimes seemed to the public that decisions were made based on limited information (especially in the beginning of the pandemic) or inconsistently. It could be said that this pandemic was a crash course into pharmacoepidemiology for the general public and some of these learnings could be put into good use with improved health data systems and models.
Expanding innovation

Probably the most common secondary use of health data today is research. Better access to registries and other health data sources enables new research and innovation.
Secondary use of health data can be used to expand development of medicine and health technology and to reduce research risks. Cheaper and easier access to data would allow new market entrants and encourage start-ups to collaborate with existing organizations. On the other hand, secondary use of health data would also strengthen health technology assessment (HTA) and to improve the pricing and reimbursement processes.
Access to large health data sets can be used to facilitate predictive modeling which would reduce the need of performing clinical trials and other expensive and slow testing.
Challenges
As already mentioned above, the key challenges related to secondary use of health data are lack of common framework and the lack of legislation. In most countries the regulatory environment does not fully support the secondary use of health data.
Because we are talking about sensitive data that is already heavily regulated, there needs to be working policies in place before the implementation can truly start.
Misuse

In a situation where a state-wide EHR system has been implemented, the state’s access to the individual’s health information could be disadvantageous to a person’s state of health in the long run. Why?
If the data is extracted not only for the purpose of informing persons or decision-making, but used as a basis for measures taken against the will of the concerned individual. For example, to check whether you have been vaccinated against COVID-19 or not and then limit the freedoms of those individuals.
This type of action, even if taken as a last choice, could be perceived as a “breach of trust” and could result in persons opting out of or not opting into an electronic health record system or not giving their consent to secondary use of their health data.
How to guarantee an individual’s legitimate expectations as well as their rights and freedoms when processing personal data?
Usually stakeholders identify the advantages in sharing health data but they also have concerns related to trust, transparency, and privacy.
Healthcare inequality
It is possible that the data-driven policies and technologies could exacerbate existing healthcare inequalities due to health data poverty (= the inability for individuals, groups, or populations to benefit from a discovery or innovation due to a scarcity of data that are adequately representative.)
It has been suggested that health data poverty is a threat to global health that could prevent the benefits of data-driven digital health technologies from being more widely realized and might even lead to them causing harm.

Known barriers
In general, it is possible to see challenges in most of the opportunities and benefits that were described above if things are managed incorrectly.
The Joint Action Towards the European Health Data Space (TEHDAS), which will be discussed in more detail below, issued a report that identified in total 20 barriers for secondary use of health data and they narrowed it down to these 11 barriers in Europe which mainly apply globally:
- There are differences in governance and health data systems between different countries.
- A lack of a common interpretation of what constitutes ‘sufficient anonymisation’ to transform personal data to non-personal data.
- A lack of a common interpretation of what constitutes ‘pseudonymisation’.
- A lack of a common interpretation of what is and is not ‘secondary use’ of data.
- National laws/rules on health and research data in addition to the GDPR and similar data privacy legislation.
- European countries can set different derogations under the GDPR.
- European countries have different preferences as to the choice of legal basis for processing personal data under the GDPR.
- Health data is considered sensitive data e.g., special category data under the GDPR, and is treated differently from other types of data when it comes to health data ethics, management, and use.
- A lack of standardized data sharing agreements for products developed by private sector providers using public health data to facilitate safe data sharing and protect public investment.
- The use of different interoperability standards makes comparisons and sharing data and research results challenging.
- Poor data management procedures reduce the ability to reuse data.
GDPR and secondary use of health data
According to the General Data Protection Regulation (GDPR) the information concerning an identifiable or identified person should be processed lawfully, fairly and in a transparent manner. The time period for storing of personal data should be strictly monitored and limited.
According to EMA’s view on GDPR and secondary use of health data, personal data concerning health should include all data pertaining to the health status of a data subject which reveal information relating to the past, current or future physical or mental health status of the data subject.
GDPR has posed major challenges to the secondary research uses. In its original implementation and interpretation GDPR fails to provide a clear basis for processing personal data for secondary research purposes. The few regulatory pathways that GDPR provides lead to complex variations among EU member states. These variations add significant transaction costs and barriers to secondary research uses of data and biospecimens.
It has been acknowledged by the authorities working on GDPR that “by coupling information from registries, researchers can obtain new knowledge of great value with regard to widespread medical conditions such as cardiovascular disease, cancer and depression. On the basis of registries, research results can be enhanced, as they draw on a larger population. […] Research results obtained through registries provide solid, high-quality knowledge which can provide the basis for the formulation and implementation of knowledge-based policy, improve the quality of life for a number of people and improve the efficiency of social services.”
The issues related to GDPR and secondary use of health data should be solved quickly so that the EU can reach the research goals set forth in the Health cluster of Horizon Europe: improving and protecting the health and well-being by generating new knowledge, developing innovative solutions to prevent, diagnose, monitor, treat and cure diseases.
European Health Data Space
EU countries have an initiative to collaborate and build European Health Data Space (EHDS). The proposal for the EU regulation on the European Health Data Space was published in 2022 and the aim is to finalize the legislation in 2024 and implement it in 2025.

The general purpose of the health data space regulation and initiative is to ensure that people have increased control over their electronic health data. The regulation also aims to ensure a legal framework consisting of governance mechanisms and a secure processing environment, allowing researchers, innovators, policy-makers and regulators to access relevant electronic health data. The regulation for health data space should contribute to a single market for digital health products and services, by harmonizing rules, and so boost healthcare system efficiencies.
As of today, in some EU countries, for example Finland, there already exists a separate law on the secondary use of health and social data, which may or may not have to be amended once the regulation becomes effective.
TEHDAS
Briefly mentioned above, the Joint Action Towards the European Health Data Space (TEHDAS) is one of the priority projects for the European Commission in the period 2021 – 2025. TEHDAS develops the concept of using secondary health data to benefit research and innovation in Europe. Their goal is that by 2025, one in three countries in the EU should offer cross border access to electronic health records.
TEHDAS analyzed the barriers for secondary use of health data and mentioned the real-life impact of the barriers and possible options to overcome this barrier and create successful secondary use of health data all over the EU.

What is the current situation in the EU?
EU countries are taking measures to develop a framework for the use of secondary health data to achieve the goal of having clear rules, common standards, and practices on health data sharing and to be in the situation where one in three EU Member States offer cross border access to EHRs in the next couple of years.
So where are we now? This can be looked at from different perspectives and below are a couple of different summaries.
Overview of the situation in the 27 EU countries as assessed by eHealth Network:
- 26 provide their citizens with access to EHR data by law
- 18 indicate that data sharing of EHRs across national borders is permitted by law
- 27 have a digital health authority, with different tasks related to interoperability, security, data protection, tele-health and m-health
- 24 report that competent authorities aim to facilitate semantic and technical interoperability
Open Data Institute (ODI) that was mentioned in the beginning of the text has generated an overall score for policy on secondary use of health data based on two measures:
- What is the quality of policy activity for this component?
- What progress is being made on implementation?
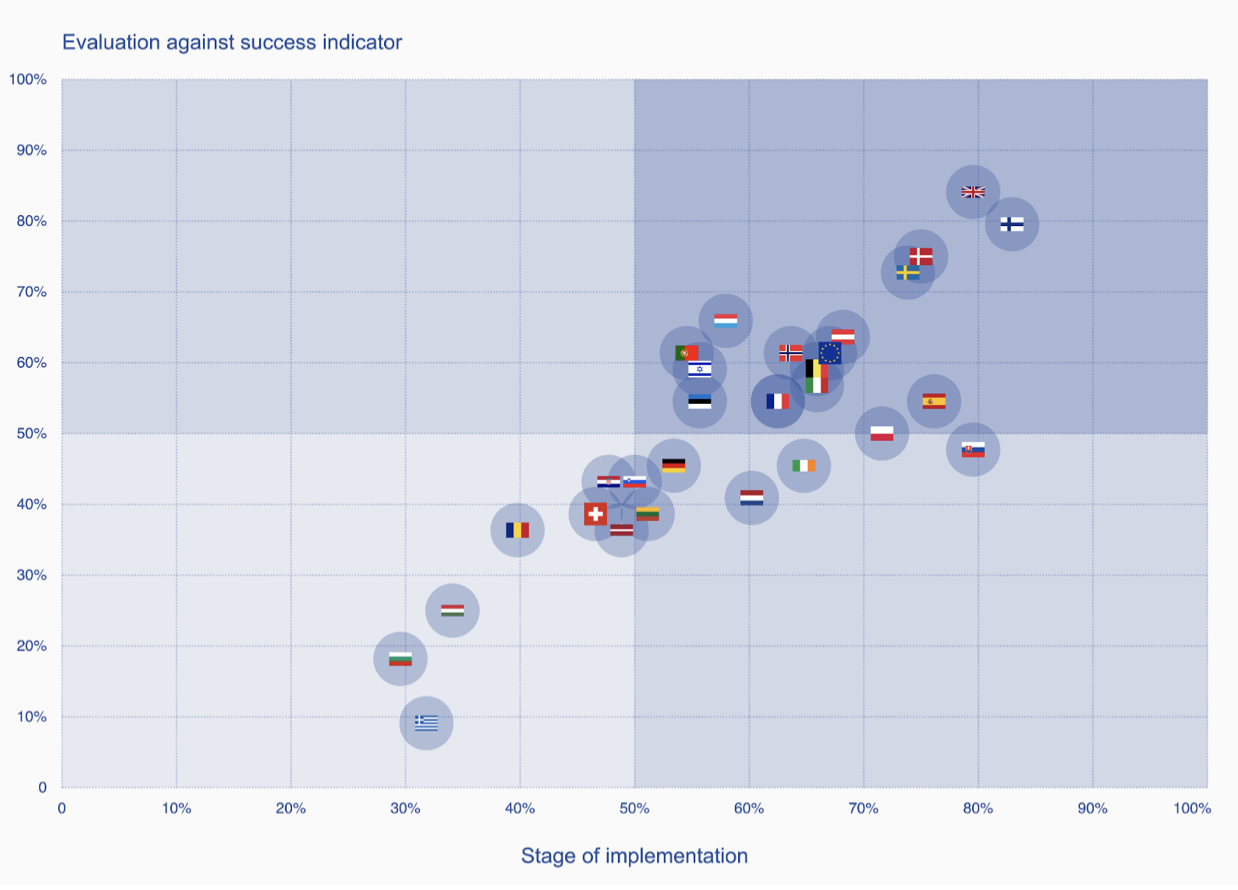
How well is your country doing on policy for secondary use of health data? ODI Tool
- United Kingdom 82%
- Finland 81%
- Denmark 75%
- Sweden 73%
- Austria 66%
- Spain 65%
- European Commission 64%
- Slovakia 64%
- Norway 63%
- Belgium 63%
- Luxembourg 62%
- Italy 61%
- Poland 61%
- France 59%
- Czechia 59%
- Portugal 58%
- Israel 57%
- Ireland 55%
- Estonia 55%
- Netherlands 51%
- Germany 49%
- Slovenia 47%
- Croatia 45%
- Lithuania 45%
- Switzerland 43%
- Latvia 43%
- Romania 38%
- Hungary 30%
- Bulgaria 24%
- Greece 20%
ODI and Roche put the European countries into the following groups
Leaders: countries where the quality of policy is stronger and the stage of implementation is more advanced.
Limited energy: countries where the quality of policy is stronger but the stage of implementation is less advanced.
Limited vision: countries where the quality of policy is weaker but the stage of implementation is more advanced.
Less prepared: countries where the quality of policy is weaker and the stage of implementation is less advanced.

Conclusion
Health being one of the key aspects of our lives, it is not surprising that health data makes up to one third of all the data generated today. Unfortunately health data is mostly unstructured and unused which means that it is still hard to use without data manipulation.
As usual with every decision, there are pros and cons related to health data policy making.
It should be clear that there is a lot of potential when it comes to secondary use of health data. The ideas listed above are not hard to grasp even if one doesn’t have a deep understanding of healthcare systems or data management.
The flip side is that data security issues are part of our everyday lives and most of us have experienced some type of data theft or misuse of our personal data. Storing even more data with even more parties can seem like a bad idea.
Some of the main questions are:
What is the value of your data?
How many of us are willing to share their health data when the time comes?
How can we coordinate global data frameworks when there is a clear lack of capabilities for international decision making and this seems to be challenging even within the US and EU?
Remedybytes 2024 | Privacy policy